Automate and centralize financial operations. The system connects production and inventory with accounting for accurate, real-time financial reporting.
Key Features:
• General ledger, AR, AP, cash, bank, budgeting
• Multi-currency support
• Financial consolidation across plants
• Automatic postings from inventory and production
One ERP. Every Industry. Global Presence. Powered by Azaalea AI

One ERP. Every Industry. Global Presence. Powered by
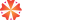
ERP for Pharmaceutical
Industry
eresource Bpro, An ERP system for Pharmaceutical, Bulk Drug & API Manufacturers
ERP for Pharmaceutical Industry
eresource Bpro, An ERP system for Pharmaceutical, Bulk Drug & API Manufacturers
Pharmaceutical Manufacturing ERP Built
for Regulatory Precision
The pharmaceutical industry operates under strict global regulations.
Manufacturers manage:
- API synthesis
- Bulk drug production
- Formulation development
- Granulation and blending
- Compression and encapsulation
- Sterile production
- Audit trails and compliance
- Injectables and syrups
- Validation protocols
- Equipment sterilization
- Controlled documentation
- Stability studies
- Quarantine and release management
Manual systems create serious risk. Batch deviations occur. Documentation errors happen. Traceability gaps delay approvals. Compliance failures lead to penalties.
eresource Bpro ERP for Pharmaceutical Industry centralizes formulation control, batch manufacturing, validation, QC testing, regulatory documentation, warehouse management, and finance into one compliant pharmaceutical ERP system.
It ensures full traceability from raw materials to final drug dispatch.
Trusted by Leading Companies




Enquire Now
Fill out the form below to get in touch with us
Why Choose eresource Bpro ERP for Pharmaceutical Manufacturing?
Pharmaceutical production requires zero tolerance for deviation. Every batch must be uniform. Every process must be validated. Every document must be audit-ready.
eresource Bpro ERP ensures structured, validated, and secure manufacturing control.
Formulation & Batch Recipe Management
Control formulas with version tracking and approval workflows.
Electronic Batch Manufacturing Records (eBMR)
Replace manual documentation with digital batch records.
API & Raw Material Traceability
Track APIs, excipients, intermediates, and finished goods by lot.
Quality Control & Stability Management
Record lab tests, assay results, stability studies, and validation data.
Regulatory Compliance Control
Support FDA, WHO-GMP, EU-GMP, MHRA, Schedule M, and 21 CFR Part 11 compliance.
Quarantine & Warehouse Management
Manage restricted zones, FEFO, temperature control, and material release.
Deviation & CAPA Tracking
Monitor production deviations and corrective actions.
As a result, pharmaceutical companies reduce risk and ensure regulatory readiness.
Core Modules
Formulation & Recipe Management
Manage every aspect of your product formulas with precision. This module helps process industries create, track, and optimize formulations while maintaining strict control over ingredients, variations, and cost impact.
Key Features:
• Multi-level formula creation with percentage or weight/volume inputs
• Alternate raw materials and potency-based adjustments
• Formula scaling for any batch size
• Revision control for formulation changes
• Restricted access for sensitive formulas
• Automatic yield, loss, overhead, and cost calculations
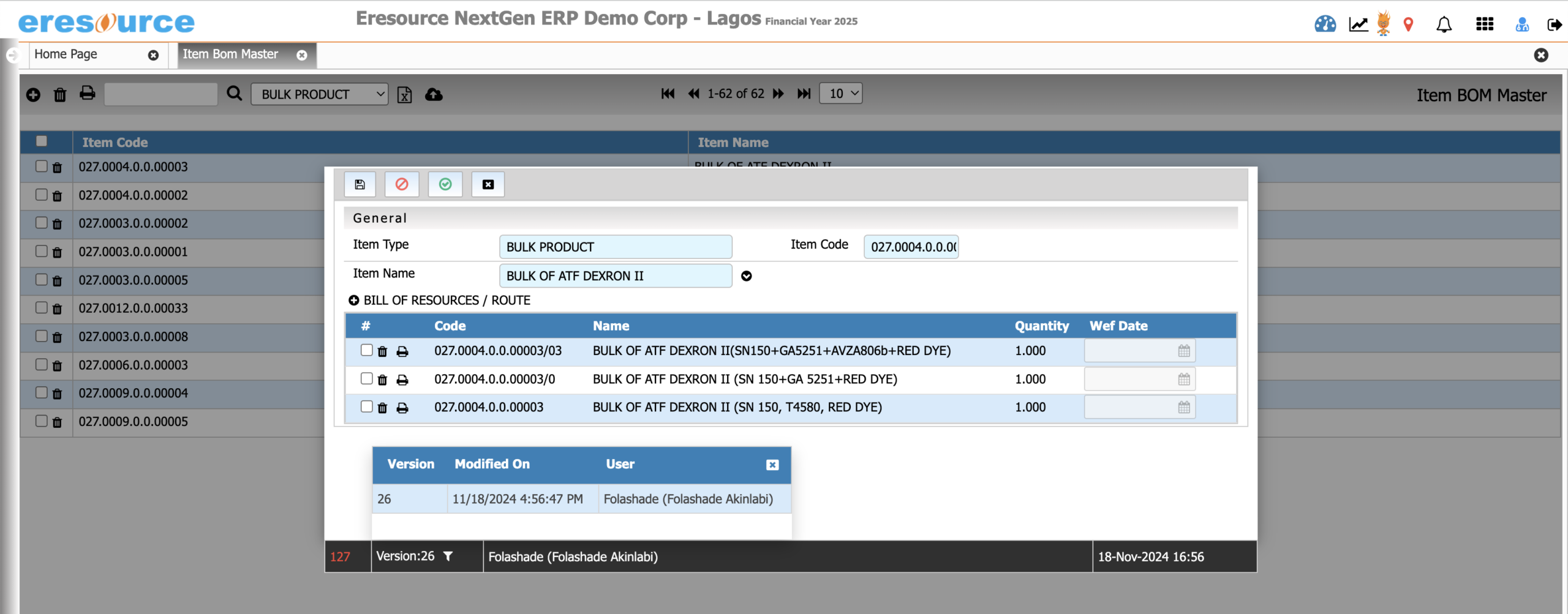
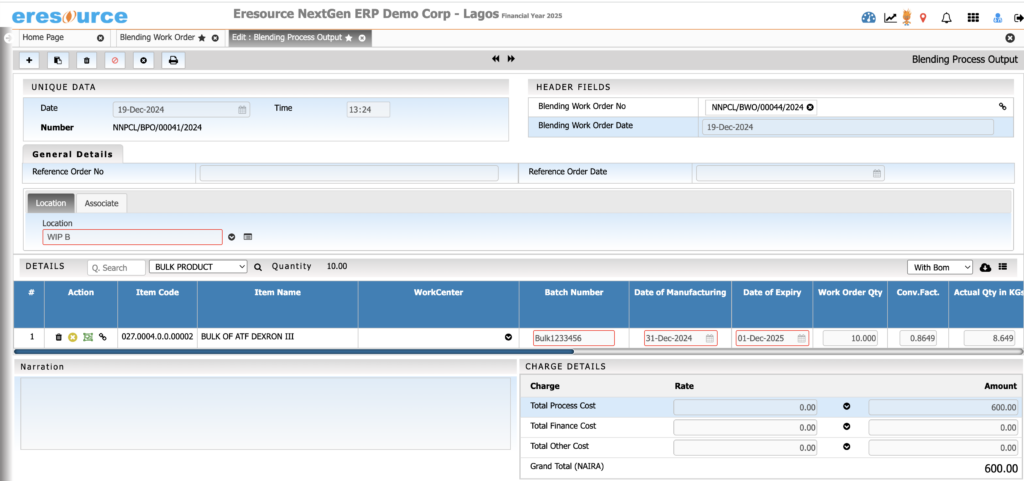
Batch Manufacturing Process
Execute batch production with complete accuracy and visibility. This module supports end-to-end batch operations and ensures compliance with regulatory requirements through detailed BMR/BPR documentation.
Key Features:
• Batch creation, scheduling, and execution
• Digital BMR and BPR tracking
• Real-time monitoring of process stages
• WIP visibility across all production points
• Auto-consumption of materials based on recipes
• Batch yield, variance, scrap, and rework recording
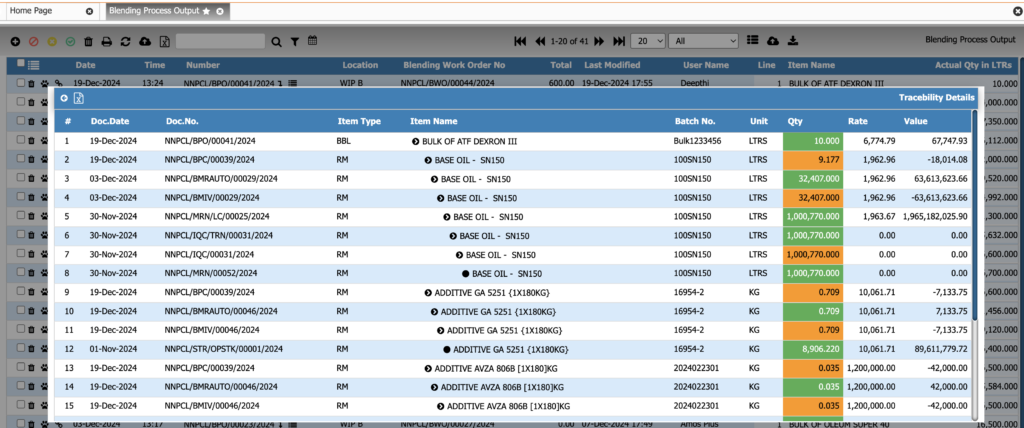
Batch Numbering & Traceability
Track every batch at every stage. This module provides full traceability from raw materials to finished products, ensuring compliance, safety, and rapid recall management.
Key Features:
• Automatic or manual batch number assignment
• Forward and backward traceability
• Complete material-to-finished-good genealogy
• Batch splitting, merging, and reclassification
• Compliance-ready traceability for pharma and food
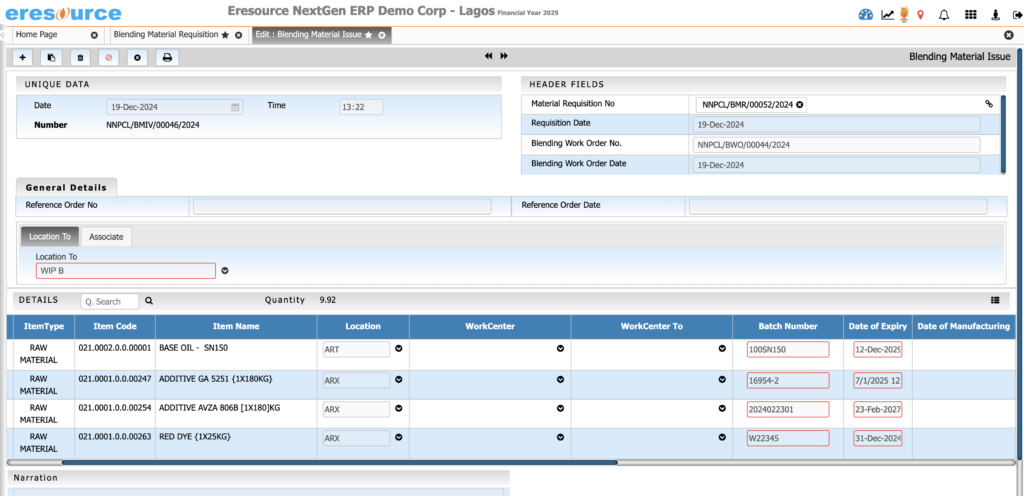
Shelf-Life & Expiry Management
Ensure material freshness and compliance by actively managing expiry timelines. The system prevents the use of expired items and improves quality control during storage and dispatch.
Key Features:
• Automated expiry and retest date calculation
• FEFO/FIFO picking
• Alerts for near-expiry materials
• Quarantine and block controls for expired items
• Automatic restrictions during production or dispatch
Batch Costing
Understand the true cost of every batch. This module delivers detailed costing insights that help manufacturers improve pricing decisions, reduce losses, and maximize profitability.
Key Features:
• Material, machine, labor, and overhead allocation
• Cost per batch, per unit, or per product variant
• Loss and wastage cost analysis
• Standard vs actual cost comparison
• Profitability tracking by batch or product line
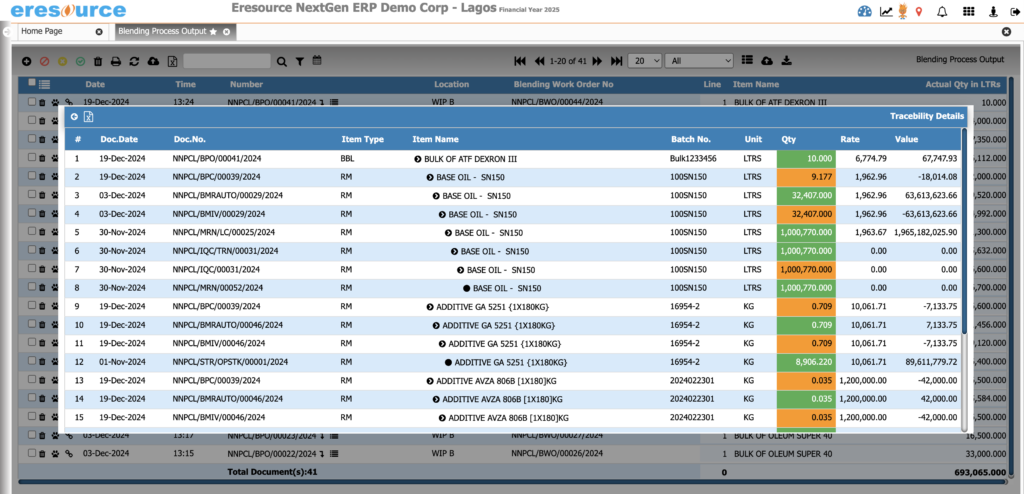
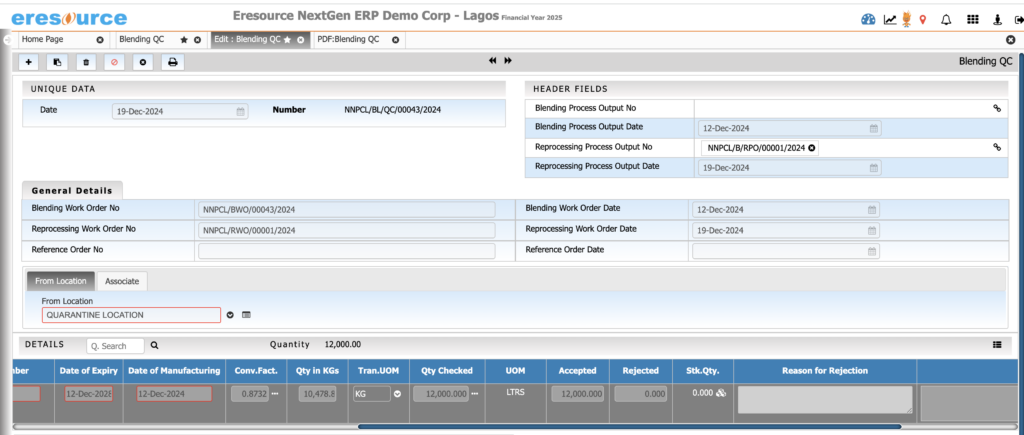
Quality Control & Quality Assurance
Ensure consistent product quality through structured QC and QA processes. From raw materials to finished goods, the system enforces strict checks, documentation, and compliance workflows.
a) Inward Quality Control (IQC)
- QC parameters for each raw material
- Sampling plans
- Hold, pass, and reject controls
- Vendor-wise QC performance
b) In-Process Quality Control (IPQC)
- Real-time QC at every batch stage
- Viscosity, pH, density, titration tests
- Auto-restriction if QC fails
- Deviation tracking and corrective actions
c) Finished Goods QC (FGQC)
- QC testing before packaging
- COA generation
- Batch-wise retain sample management
- QA release workflows
d) Stability Testing
- Stability study scheduling
- Condition-wise test logging
- Stability-based expiry assignment
Inventory & Warehouse Management
Control your inventory with complete batch accuracy. This module ensures compliant storage, stock rotation, and material issuance for smooth and safe production cycles.
Key Features:
• Batch, grade, potency-based stock visibility
• FEFO/FIFO allocations
• Approved, rejected, and quarantine stock segregation
• Raw material issuance based on recipe
• Warehouse transfers with batch tracking
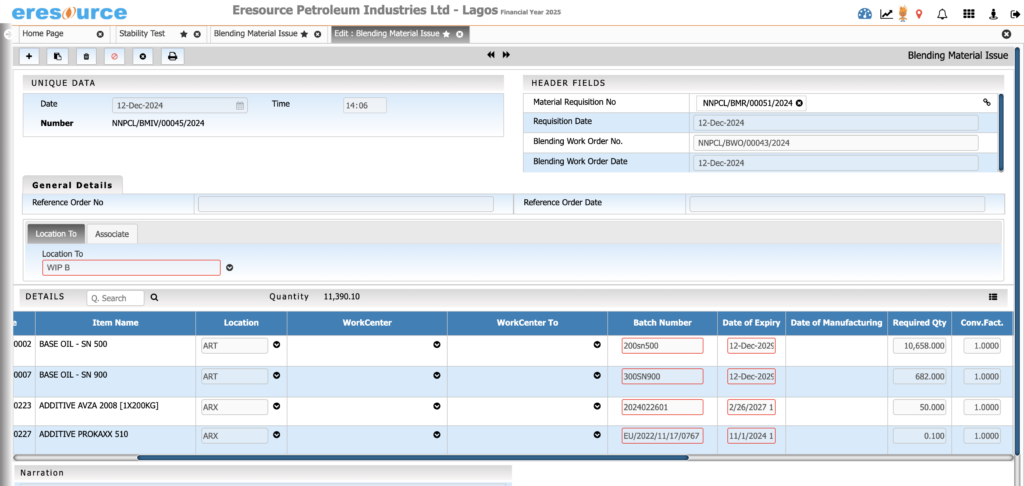
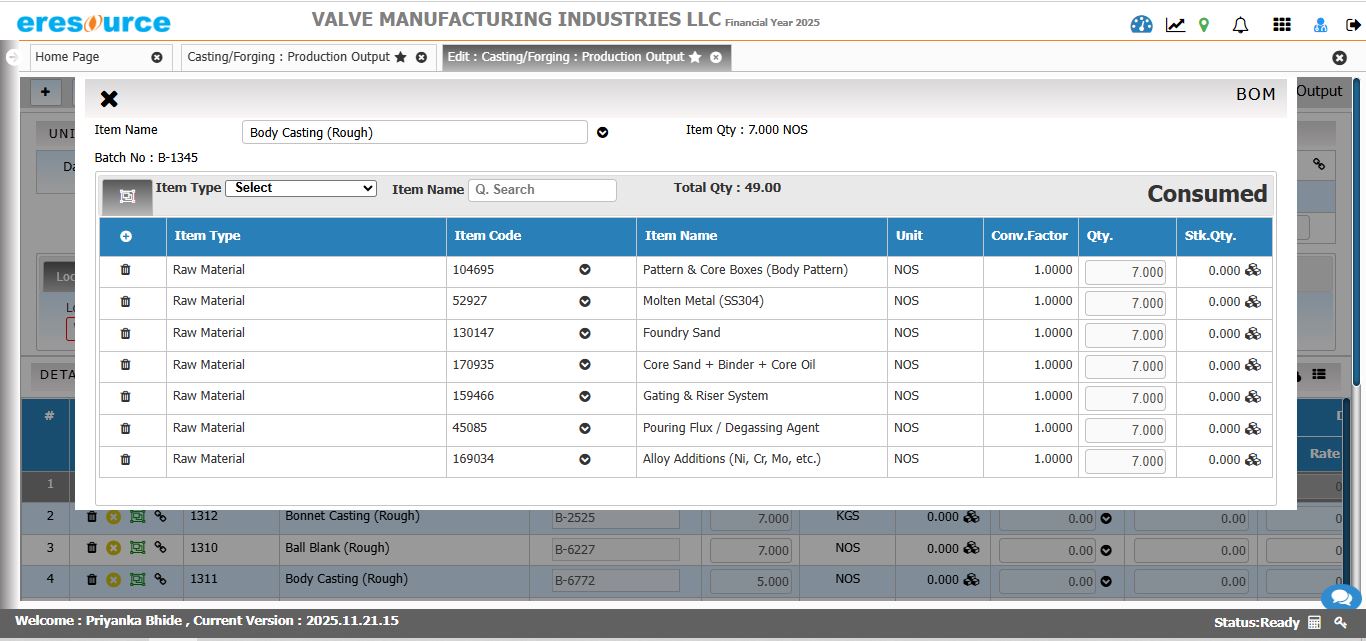
Production Planning & Scheduling
Plan production efficiently with real-time insights into capacity, availability, and demand. The system optimizes batch schedules across tanks, reactors, and processes for maximum throughput.
Key Features:
• MRP-driven raw material planning
• Reactor and vessel capacity scheduling
• Multi-level batch planning
• Automated scheduling for multi-stage processes
• Resource load balancing
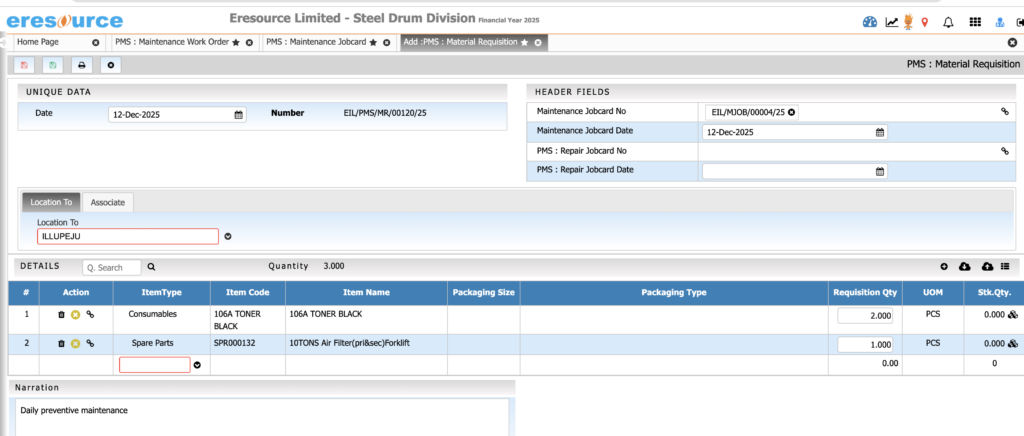
Equipment & Maintenance Management
Keep production equipment running reliably. This module reduces downtime by scheduling preventive maintenance and tracking all repairs, calibrations, and associated costs.
Key Features:
• Preventive and breakdown maintenance
• Calibration and equipment validation schedules
• Maintenance work order tracking
• Cost monitoring for maintenance tasks
• Spare parts inventory
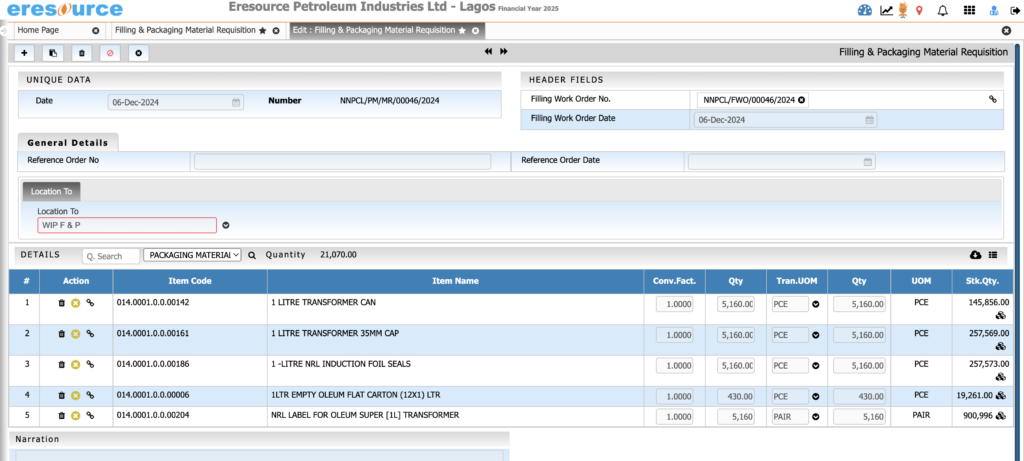
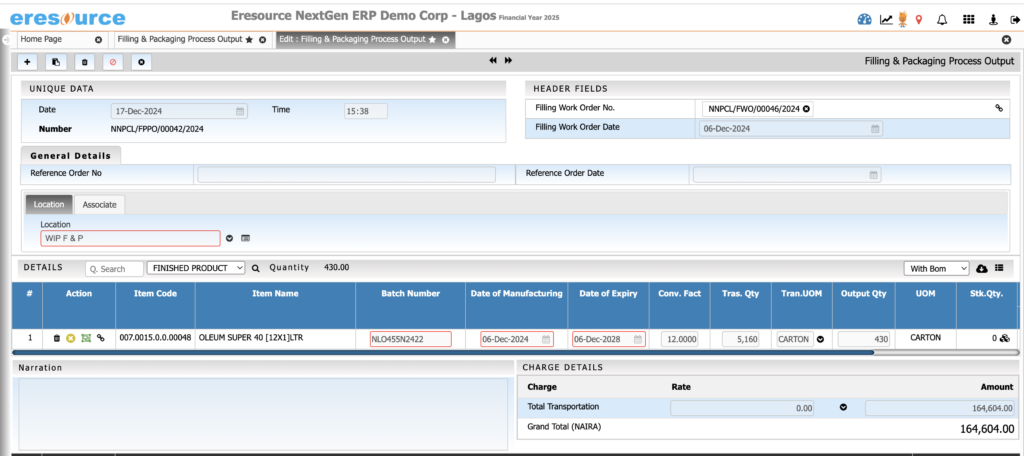
Packaging & Filling Management
Manage packaging operations seamlessly. The system supports primary and secondary packaging and ensures accurate labeling and stock deductions.
Key Features:
• Multi-stage packaging workflows
• Packaging BOM with labels, cartons, sleeves, caps
• Label printing with batch and expiry information
• Automatic consumption of packing materials
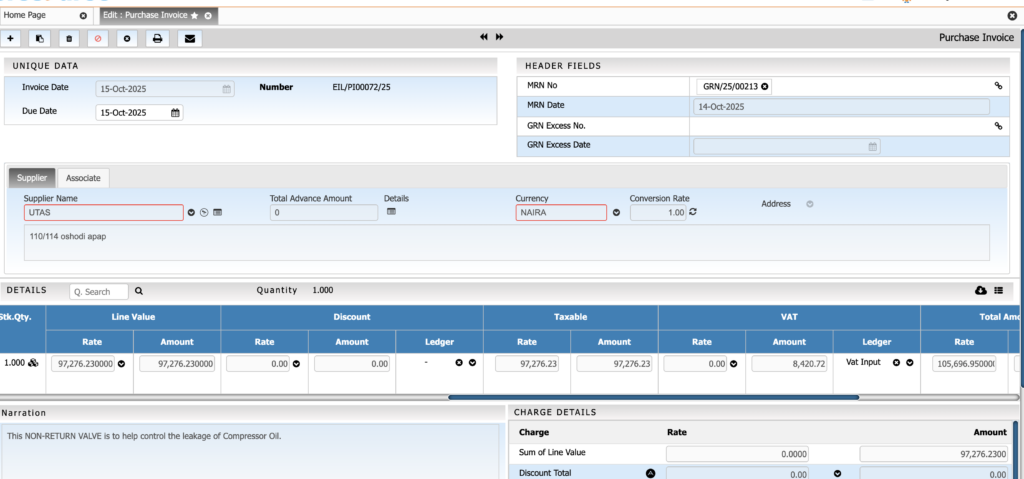
Purchase & Supplier Management
Streamline procurement with controlled and transparent purchasing processes. This module helps ensure timely availability of raw materials while maintaining vendor quality and cost efficiency.
Key Features:
• RFQ and quotation comparison
• Vendor qualification and performance tracking
• Potency-based purchase adjustments
• Purchase contracts for bulk chemicals
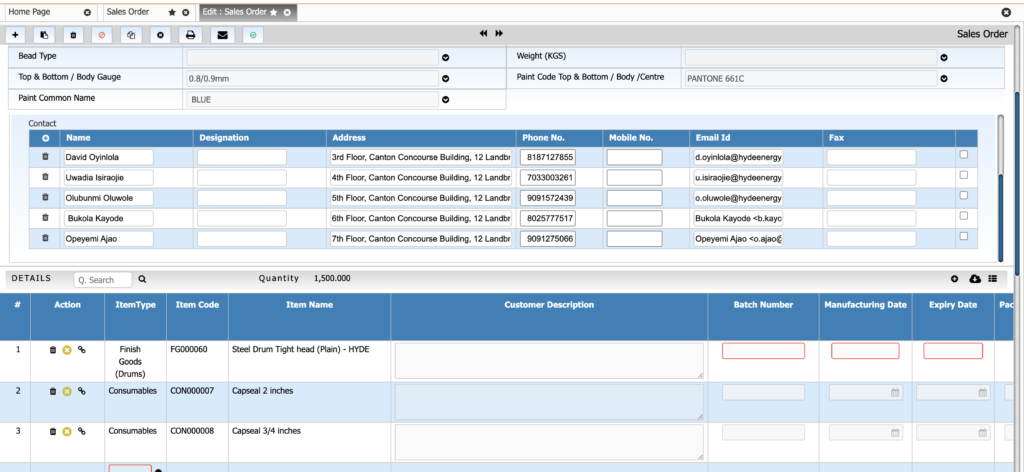
Sales & Order Management
Handle sales orders with precise control over batch selection and expiry constraints. This ensures timely dispatch and compliance with customer-specific requirements.
Key Features:
• Batch-specific order allocations
• FEFO-based batch selection
• COA attached with dispatch
• Sales forecasting linked with production
Financial Management
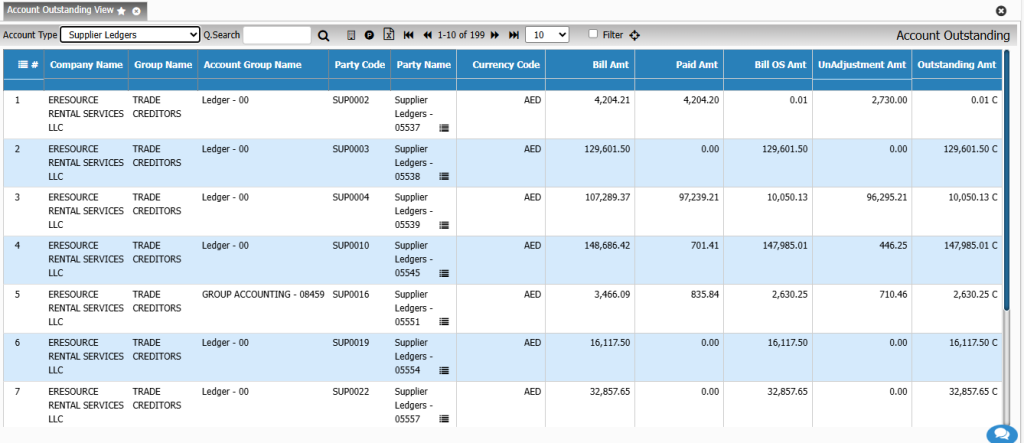
Costing & Profitability Analysis
Get a clear view of operational profitability. This module analyzes cost behavior across batches, products, and customers, enabling smarter financial decision-making.
Key Features:
• Activity-based costing
• Batch-wise cost variance
• Product profitability analysis
• Cost simulation for new formulations
Key Benefits for Pharmaceutical & API Manufacturers
- Maintain strict batch uniformity through controlled formulations
- Achieve full traceability across raw materials, APIs, intermediates, and final drugs
- Strengthen compliance with FDA, WHO, GMP, and Schedule M standards
- Reduce production deviations and manual documentation errors
- Improve warehouse safety with quarantine, FEFO, and controlled storage
- Enhance costing accuracy with real-time batch consumption tracking
- Increase efficiency with automated workflows and digital batch records
- Scale across multiple dosage forms and multi-plant operations
Built for Pharmaceutical & Bulk Drug Manufacturers

- Bpro ERP supports:
- API manufacturers
- Bulk drug producers
- Formulation manufacturing units
- Tablet and capsule manufacturers
- Injectable and sterile units
- Syrup and suspension manufacturers
- Contract manufacturing organizations (CMOs)
- Multi-plant pharmaceutical companies
Unlike generic ERP software, Bpro ERP is designed for highly regulated pharmaceutical manufacturing environments.
How Pharmaceutical ERP Improves Compliance & Profitability
Pharmaceutical companies often struggle with:
- Manual documentation errors
- Audit findings
- Batch rejection
- Deviation management
- Regulatory inspection pressure
- Inventory expiry losses
However, with eresource Bpro ERP:
- Every batch is digitally recorded
- Every validation step is logged
- Every material lot is traceable
- Every deviation is tracked
- Every compliance document is audit-ready
Therefore, inspection readiness improves and compliance risk reduces.
Advanced Manufacturing Capabilities
Electronic Batch Records (EBR)
Maintain validated digital batch documentation.
Audit Trail Management
Track user activity and data changes securely.
Validation & Qualification Tracking
Monitor equipment validation and calibration schedules.
Controlled Access & Data Integrity
Ensure secure role-based system access.
Case Studies
MediCore Pharmaceuticals
Reduced batch release time by 20% through automated electronic batch record workflows and integrated QC testing.
20% Faster Batch Release
PharmaMax Ltd
Achieved zero audit issues in recent FDA inspection with comprehensive compliance documentation and traceability.
Zero Audit Issues
API Integrations
APIs to integrate with other enterprise, compliance, finance, analytics, or planning systems as per business needs.
Real-time data exchange with LIMS, regulatory reporting systems, serialization platforms, and quality management systems.
IoT Integrations
Cold storage monitoring for temperature-sensitive materials
RFID tracking for complete batch genealogy
Cleanroom environmental sensors for GMP compliance
Video Resources
Play Video
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Play Video
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Play Video
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Play Video
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Frequently
Asked Question
Get answers to common questions about Pharmaceutical Industry

Can eresource Bpro handle formulation and master recipe management?
Yes. It manages detailed formulations, ingredient ratios, and validated master recipes.
Is the ERP compliant with FDA, GMP, WHO, and Schedule M guidelines?
Absolutely. It supports regulatory documentation, audit trails, and validation workflows.
Can it manage APIs, intermediates, and controlled raw material storage?
Yes. It supports full traceability, quarantine, FEFO, and controlled warehouse access.
Does the system support QC activities like stability and microbial tests?
Yes. All lab tests, stability records, and inspection logs are captured digitally.
Can it track granulation, compression, coating, and filling processes?
Yes. Eresource Bpro tracks each production stage with complete WIP visibility.
Is Bpro suitable for multi-plant and large-scale pharma manufacturing?
Yes. It is built for both small units and large multi-location pharmaceutical companies.
Transform Pharmaceutical Manufacturing with eresource Bpro
Experience how eresource Bpro ERP simplifies formulation, batch production, quality testing, regulatory compliance, and financial management.
